Breast cancer, while it’s a hard burden to bear, can usually be treated in countries like the United States, Sweden or Japan, albeit, at a great cost. But, in the case of developing countries, the situation is completely different and the low survival rates registered there are usually the result of a lack of early detection programs.
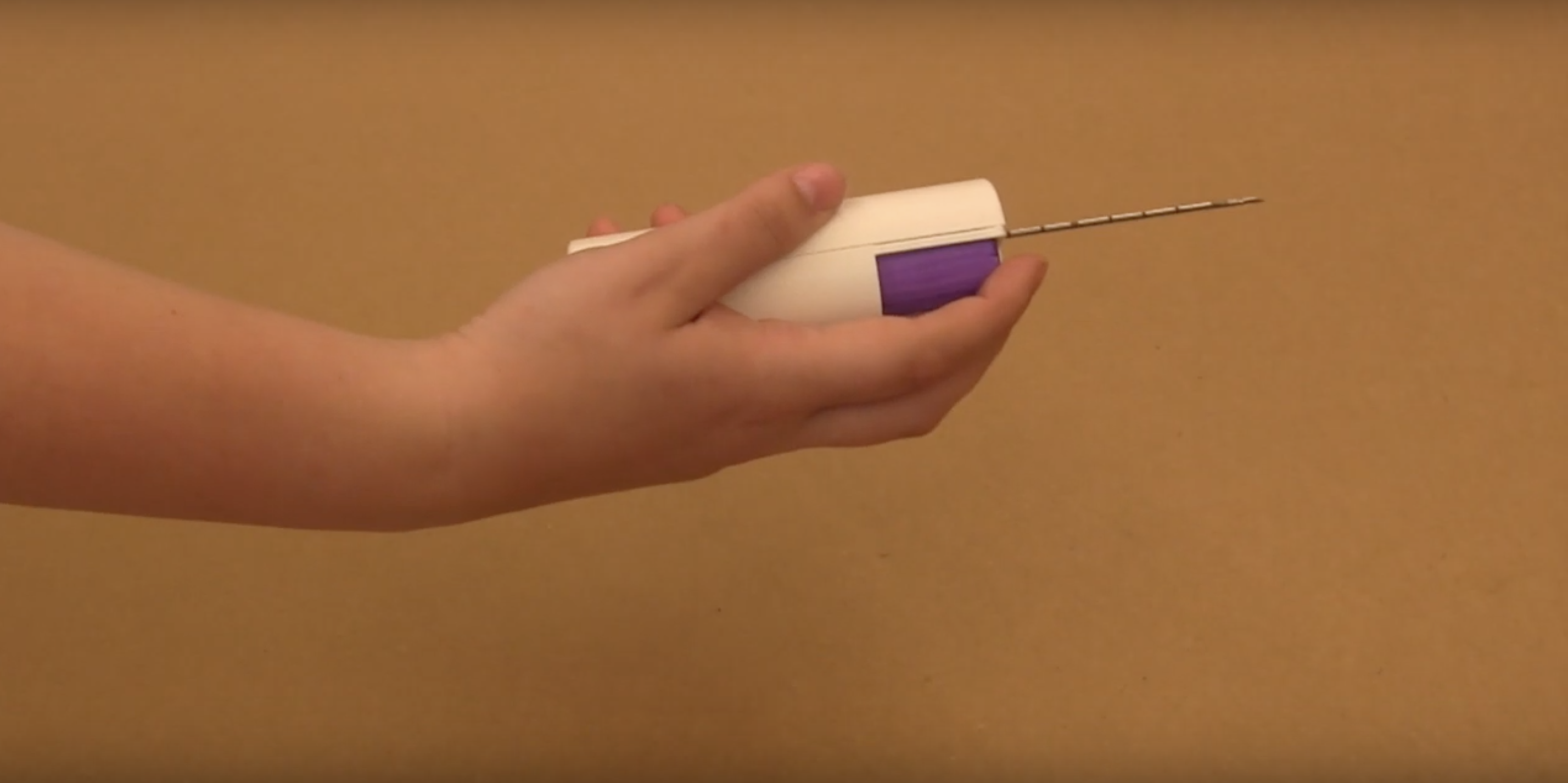
However, four female students at the Johns Hopkins University have developed a low-cost, disposable needle biopsy technology that might just be able to help the doctors and nurses screen their patients without raking up the costs or even the waste.
The device is called Ithemba, which is a word from Swahili, meaning ‘hope’ and the women have been working on it since three years ago.

The Ithemba team: Laura Hinson, Madeline Lee, Valerie Zawicki and Sophia Triantis
“We’ve designed a novel, disposable portion that attaches to the reusable device and the disposable portion has an ability to trap contaminants that would come back through the needle into the device,” said Sophia Triantis in an interview with TechCrunch “What we’ve created is a way to trap that and have that full portion be disposable and making the device as easy to clean as possible… with a bleach wipe.”
A mechanism inside the device prevents the blood from being pulled back into the needle, which makes the reusable needless less likely to be contaminated and a far better alternative to the expensive, disposable needles that the clinics and hospitals in developing countries simply cannot afford.
Unfortunately, it will take time until we’ll be able to see it in action: according to undergraduate student Valerie Zawicki, we’ll see the device on the market in maybe three to five years. But potential partnerships with big companies might cut that time in half and already a clinic in South Africa and a hospital in Peru have expressed their interest in the Ithemba.
The four women hope that, if all goes well and Ithemba gets to be certified as a Class 2 medical device, they can begin clinical trials as early as the summer of 2020.
Also Read:✍️ MIT AI Model Can Detect Breast Cancer Years in Advance✍️
Follow TechTheLead on Google News to get the news first.